Proof that Ignites Progress

Together we are building a comprehensive body of evidence to drive innovation and clinical excellence
MOMENTIS Study of the MITRIS RESILIA Valve: One of the Largest Core-Lab Adjudicated Surgical Mitral Valve Replacement (MVR) Studies
Results at 1 year
An ongoing prospective, observational, post-market, single-arm, real-world evidence study in 351 North American adults with a dysfunctional native or prosthetic mitral valve who required replacement with a MITRIS RESILIA Mitral Valve.1
Freedom from valve-related death
Freedom from valve-related reintervention
For more details, download the clinical infographic.

At the 1-year follow-up, the MOMENTIS study demonstrated promising results for real-world mitral valve patients with the MITRIS RESILIA mitral valve.
MOMENTIS study follow-up is ongoing and will continue through 10 years

COMMENCE Mitral Trial: Early Clinical Data Supporting RESILIA Tissue in the Mitral Position
Results at 5 years
Mid-term outcomes of the ongoing COMMENCE mitral IDE trial of 82 patients who underwent MVR in this prospective, single-arm trial.2
Freedom from all-cause mortality
Freedom from SVD
Freedom from reoperation
For more details, download the clinical summary.
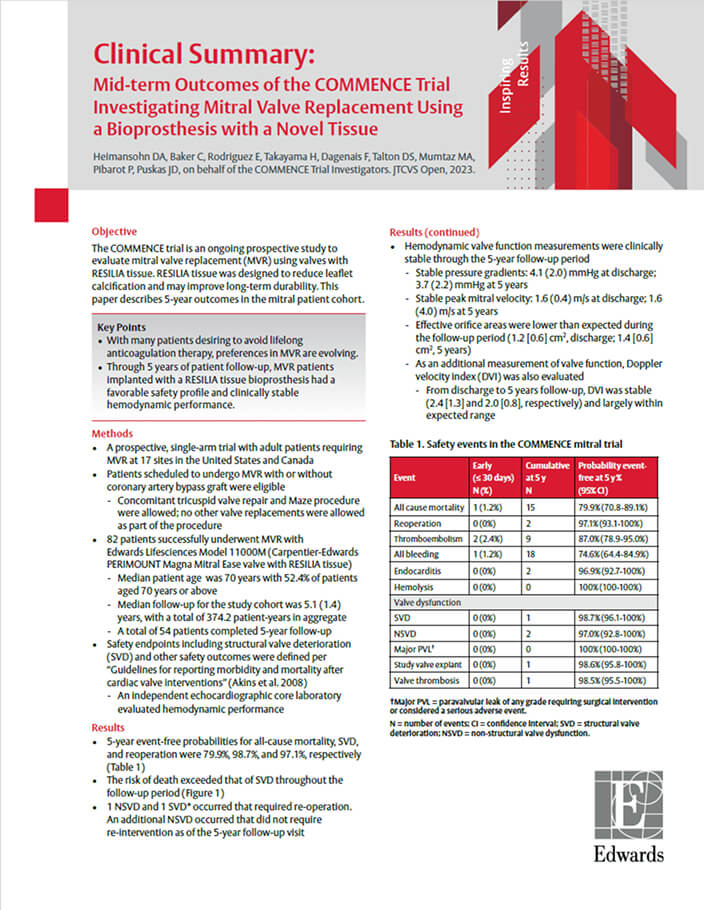
With a favorable safety profile and clinically stable hemodynamic performance, this study supports the use of RESILIA tissue in the mitral position with excellent durability out to 5 years.
RESILIA tissue valves in the mitral position offer excellent durability out to 5 years

References
- Ailawadi G, Heimansohn D, Accola K, et al. One-year safety and performance of a novel bovine pericardial mitral valve with advanced anti-calcification treatment. Presented at the 2026 Society of Thoracic Surgeons Annual Meeting. January 29, 2026.
- Heimansohn DA, Baker C, Rodriguez E, et al. Mid-term outcomes of the COMMENCE trial investigating mitral valve replacement using a bioprosthesis with a novel tissue. JTCVS Open. 2023;15:151-163. Published 2023 Jun 2. doi:10.1016/j.xjon.2023.05.008
- Akins CW, Miller DC, Turina MI, et al. Guidelines for reporting mortality and morbidity after cardiac valve interventions. Ann Thorac Surg. 2008;85(4):1490-1495. doi:10.1016/j.athoracsur.2007.12.082
Important Safety Information
RESILIA Tissue Devices
Indications: INSPIRIS RESILIA Aortic Valve - For use in replacement of native or prosthetic aortic heart valves. KONECT RESILIA Aortic Valved Conduit - For use in replacement of native or prosthetic aortic heart valves and the associated repair or replacement of a damaged or diseased ascending aorta. MITRIS RESILIA Mitral Valve - For use in replacement of native or prosthetic mitral heart valves.
Contraindications: There are no known contraindications with the use of these RESILIA tissue heart valve devices.
Complications and Side Effects: INSPIRIS RESILIA Aortic Valve: Thromboembolism, valve thrombosis, hemorrhage, hemolysis, regurgitation, endocarditis, structural valve deterioration, nonstructural dysfunction, stenosis, arrhythmia, transient ischemic attack/stroke, congestive heart failure, myocardial infarction, any of which could lead to reoperation, explantation, permanent disability, and death. Additional adverse events potentially associated with the use of polyester vascular grafts in the KONECT RESILIA AVC include hemorrhage, thrombosis, graft infection, embolism, aneurysm, pseudoaneurysm, seroma, occlusion (anastomotic intimal hyperplasia), immunological reaction to collagen (shown to be a weak immunogen; infrequent, mild, localized and self-limiting), intimal peel formation, and conduit dilatation. MITRIS RESILIA Mitral Valve - Thromboembolism, valve thrombosis, hemorrhage, hemolysis, regurgitation, endocarditis, structural valve deterioration, nonstructural dysfunction, stenosis, arrhythmia, transient ischemic attack/stroke, congestive heart failure, myocardial infarction, ventricular perforation by stent posts, any of which could lead to reoperation, explantation, permanent disability, and death.
Warnings: INSPIRIS RESILIA Aortic Valve - DO NOT ADJUST THE VALVE DIAMETER BY EXPANDING THE BAND PRIOR TO OR DURING IMPLANTATION OF THE SURGICAL VALVE. The expandable band is not designed to allow for compression or expansion during implantation of the surgical valve. This will cause damage to the valve and may result in aortic incompetence. DO NOT PERFORM STAND-ALONE BALLOON AORTIC VALVULOPLASTY PROCEDURES ON THIS VALVE FOR THE SIZES 19 - 25 mm as this may expand the valve causing aortic incompetence, coronary embolism or annular rupture. Valve-in-valve sizing in the INSPIRIS valve has only been tested with specific Edwards transcatheter heart valves. Use of other transcatheter valves may result in embolization of transcatheter devices anchored within or result in annular rupture.
CAUTION: US law restricts these devices to sale by or on the order of a physician. See instructions for use for full prescribing information.
