freedom from SVD
95% CI: 96.0, 99.8
Now is the time to consider RESILIA tissue for your patients

The latest European guidelines state that mechanical aortic valves should be considered in patients aged <60 years, but valve choice depends on a variety of factors, including patient preference, patient life expectancy, and expected prosthetic valve durability.6
Choosing a mechanical valve means a lifetime of anticoagulant therapy that brings with it lifestyle restrictions and an elevated risk of major bleeding. There are dietary considerations, potential drug-drug interactions, ongoing international normalized ratio monitoring, and frequent doctor visits.2,6
Recipients of tissue valves do not always need long-term anticoagulation therapy, so are not always impacted by the dietary restrictions and lifestyle limitations associated with lifelong anticoagulation.2


The Heart Team should consider patient preference when choosing a valve, alongside relevant updated evidence, key medical considerations and other lifetime management considerations.6


The INSPIRIS RESILIA valve incorporates VFit technology* for potential future valve-in-valve procedures which is not possible with mechanical valves1
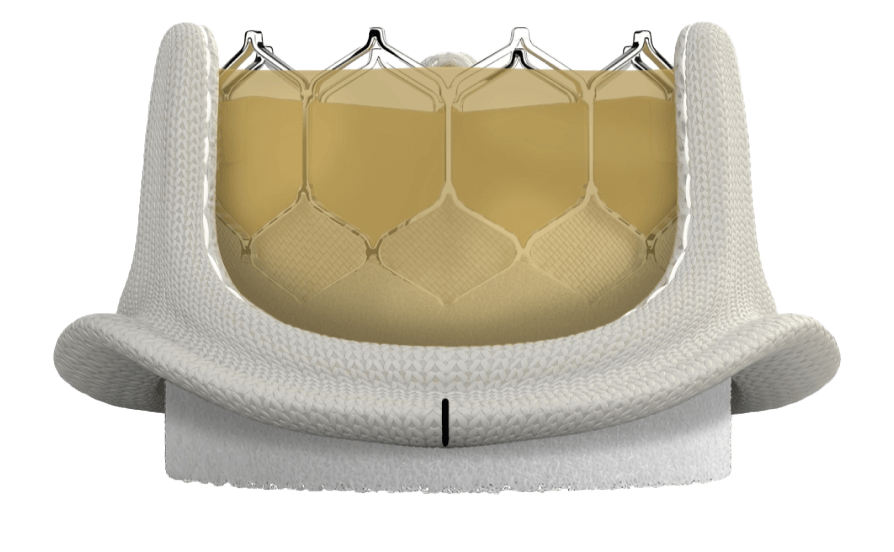
Cobalt-chromium expansion band is designed to support controlled, uniform expansion and facilitates potential future ViV transcatheter procedures without requiring high-pressure fracturing or full replacement1
*Refer to device Instructions for Use for important warnings related to VFit technology. These features have not been observed in clinical studies to establish the safety and effectiveness of the model 11500A for use in valve-in-valve procedures. VFit technology is available on sizes 19 -25 mm.

Results through 10 years5
95% CI: 96.0, 99.8
95% CI: 95.8, 99.7
Demonstrating continued leadership in surgical bioprostheses
It’s time for you and your patients to make the choice that is suitable for them. A fully informed, shared choice that gives them confidence in what is to come


For a listing of indications, contraindications, precautions, warnings, and potential adverse events, please refer to the Instructions for Use (consult eifu.edwards.com where applicable).
PP--EU-11861 v1.0
We are committed to providing your institution, clinicians and staff with the highest levels of customer service and support to ensure seamless product implementation and ongoing use, including:
Monday through Friday, 8:00am to 17:00pm CET
